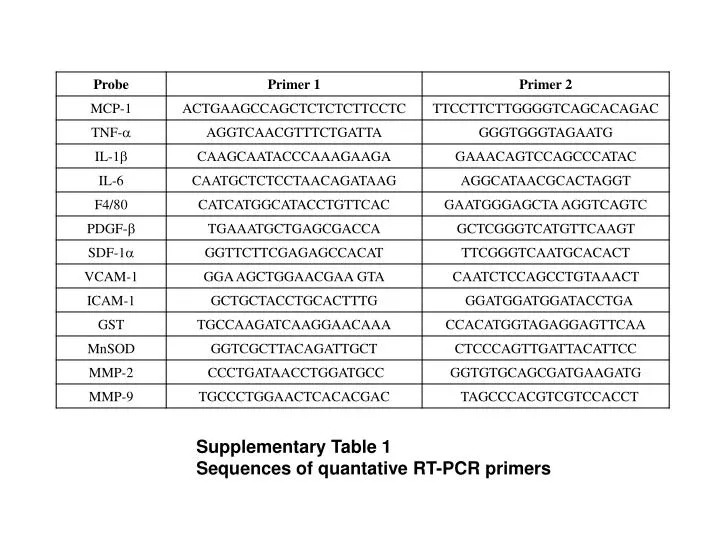
We present here a suite of primers designed to amplify Dikaryomycota efficiently, requiring little optimization for use in the hands of even relatively untrained operators. Subsequently, the plant-excluding primers ITS1-F and ITS4-B came into wide use for analyses of fungal ITS sequences, but these primers were "intended to be specific to fungi and basidiomycetes, respectively". 
These primers amplify a wide range of fungal targets and work well to analyze DNA isolated from individual organisms, but do not exclude effectively the plant host sequences in mixed, phytosphere DNA extracts typical of studies of plant-associated microbiota. The earliest PCR primers to gain wide acceptance for work with fungal Internal Transcribe Sequences (ITS) were "ITS1" and "ITS4" which amplify the highly variable ITS1 and ITS2 sequences surrounding the 5.8S-coding sequence and situated between the Small SubUnit-coding sequence (SSU) and the Large SubUnit-coding sequence (LSU) of the ribosomal operon. Of particular interest for such work is the division Dikaryomycota, which includes the subdivisions Basidiomycotina and Ascomycotina, and encompasses all ectomycorrhizal fungi. Primers allowing simultaneous analysis of all the fungal phyla which are involved in ectomycorrhizal symbioses would be a useful tool in studying the ecology of these fungi. Analysis of natural populations of ectomycorrhizas requires high rates of sampling because of the high number of fungal species involved and the high spatial variability observed in natural systems. The laborious nature of microscopic analysis and identification is the driving force behind developing such methods as molecular verification of identified morphotypes by means of PCR-RFLP. Analysis of natural ectomycorrhizal fungal communities traditionally has been a laborious, highly-skilled process with heavy reliance on gross morphological characterization of the ectomycorrhizal root-tips. For instance, the fungi forming ectomycorrhizal symbioses number over 5000 species. Studies of fungi (Kingdom Eumycota) in natural environments often require simultaneous analysis of a broad taxonomic range. The complete set of primers was developed with an emphasis on discrimination between plant and fungal sequences and should be particularly useful for studies of fungi where samples also contain high levels of background plant DNA, such as verifying ectomycorrhizal morphotypes or characterizing phylosphere communities. These plant primers have been successfully applied to PCR-RFLP analyses of forest plant tissues from above- and below-ground samples and work well at distinguishing a selection of plants to the species level. These were used to verify that the host plant DNA was not being amplified with the fungal primers. A set of wide-range plant-specific primers were developed at positions corresponding to one pair of the fungal primers. These primers have also been successfully applied to Quantitative PCR (QPCR), Length Heterogeneity PCR (LH-PCR) and Terminal Restriction Fragment Length Polymorphism (T-RFLP) analyses of fungi. Single ectomycorrhizal root tips were similarly analyzed. A range of basidiomycete fruiting bodies and ascomycete cultures were analyzed with the nested set of primers and Restriction Fragment Length Polymorphism (RFLP) fingerprinting to demonstrate the specificity of the assay. Primers in the 5.8S sequence were also developed that would permit separate amplifications of ITS1 and ITS2. A nested set of 4 primers was developed that reflected these criteria and performed well amplifying ITS regions of fungal rDNA. Numerous sequences for PCR primers were tested to develop PCR assays with a wide range of fungal compatibility and high discrimination from plant DNA. Typically, it has been necessary to use multiple primer sets to accommodate the range of fungi under study, potentially creating artificial distinctions for fungal sequences that amplify with more than one primer set. Previously published PCR primers available for amplifying these sequences from environmental samples provide varying degrees of success at discriminating against plant DNA while maintaining a broad range of compatibility. The Internal Transcribed Spacer (ITS) regions of fungal ribosomal DNA (rDNA) are highly variable sequences of great importance in distinguishing fungal species by PCR analysis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
